And don’t forget to put the unit g/mol to your final calculated molar mass.It’s a periodic table with names, element symbols, atomic numbers, atomic weights, and groups. You can see that in ZnCl2, there is 1 Zinc atom and 2 Chlorine atoms. This is your go-to free black and white printable periodic table for facts and figures. 2 Now, to calculate the molar mass of ZnCl2, you just have to add the molar mass of all the individual atoms that are present in ZnCl2. The element names are as large as I could consistently make them. The molar mass of Chlorine is 35.45 g/mol. This periodic table includes all 118 elements known as of 2017. First solve the brackets, then multiplications and at last do the final addition. This color periodic table contains the usual element numbers, symbols, element names, and atomic masses, but has the added bonus of larger type. 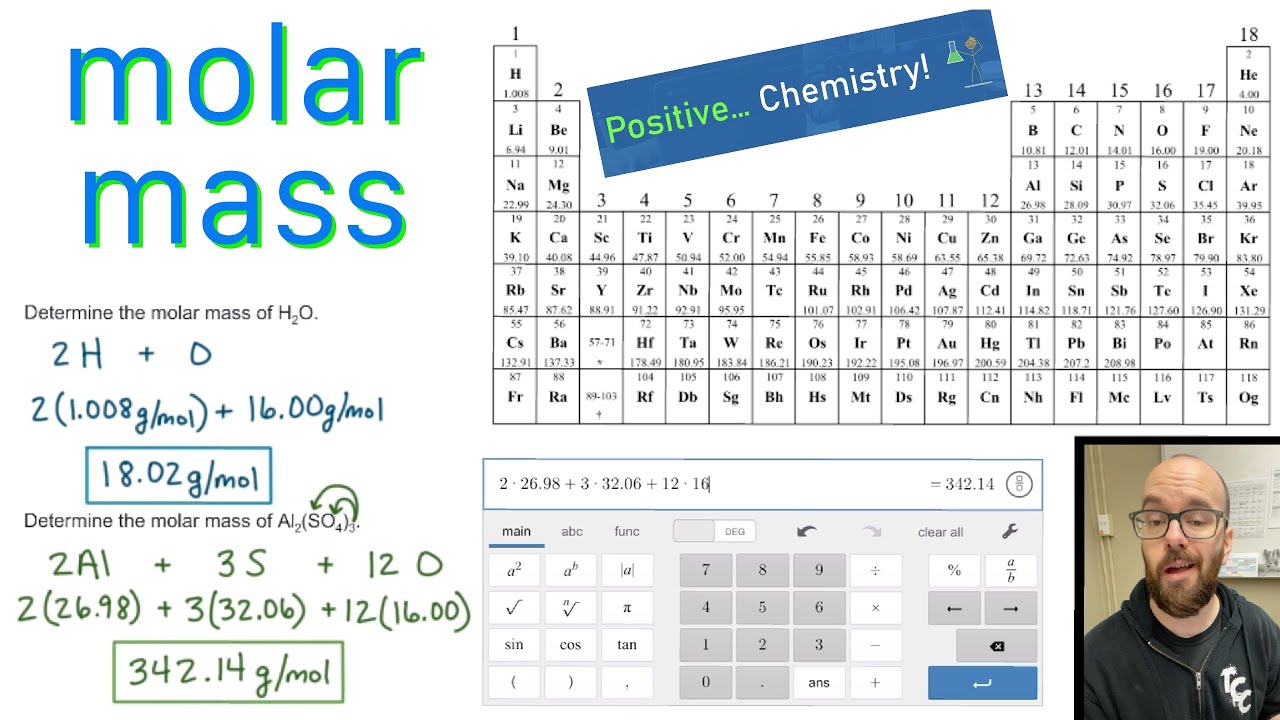
It can supply you with an exact representation of a substance’ssize and mass, and valence electron shell. You can see that The molar mass of Sodium is 22.990 g/mol. Periodic Table With Atomic And Molar Mass The Periodic Dinner table is an essential part of the research into scientific research, also it can be helpful in figuring out a substance’s attributes. So let’s look at the molar mass of Sodium, Sulfur and Oxygen from the above periodic table. Now in Na2SO4, there are 2 Sodium atoms, 1 Sulfur atom and 4 Oxygen atoms. Always follow the calculation order to avoid any mistakes in calculation. You can see the molar mass value of all the atoms from this periodic table.But all these units (i.e g/mol, grams/mole and g/mole) are the same. There is also a 2019 edition of this table. Here is the pdf file of the color periodic table so you can save and print it. It includes element names, symbols, atomic numbers, atomic weights, element groups, and periods. In some books, you may see the unit of molar mass as grams/mole or g/mole. Periodic Table of Elements with Relative Atomic Masses Periodic Table Key X Synthetic Elements X Liquids or melt at close to room temp. 2013 Edition This free periodic table wallpaper has a white background.I hope you have understood the short and simple calculation for finding the molar mass of Na2O. This is a periodic table with atomic mass, element name, element symbol, and atomic number.The atomic mass is the average number of protons and neutrons in atoms of a chemical elements, allowing for the natural abundances of the element’s isotopes. It is a constant property of each substance for example, the. Molar mass is the mass of 1 mole of the solute. We can link the number of entities X in a specified sample N(X). Hence the Molar mass of Na2O is 61.979 g/mol. Moles allow you to directly read weight from the periodic table (e.g., 1 mole of N is 28 g or 1 mole of NaCl is 58.5 g). So, Molar mass of Na2O = Molar mass of 2 Sodium (Na) atoms + Molar mass of 1 Oxygen (O) atom. You can see that in Na2O, there are 2 Sodium atoms and 1 Oxygen atom. And don’t forget to put the unit g/mol to your final calculated molar mass.Now, to calculate the molar mass of Na2O, you just have to add the molar mass of all the individual atoms that are present in Na2O.The given mass of K (4.7 g) is a bit more than one-tenth the molar mass (39.10 g), so a reasonable ballpark estimate of the number of moles would be slightly greater than 0.1 mol. First solve the brackets, then multiplications and at last do the final addition. Referring to the periodic table, the atomic mass of K is 39.10 amu, and so its molar mass is 39.10 g/mol. (262) Interactive periodic table of elements - your complete guide to the elements including definition, mass & names of each chemical in the periodic table. Always follow the calculation order to avoid any mistakes in calculation.But all these units (i.e g/mol, grams/mole and g/mole) are the same. American Chemical Society 57-71 Lithium 6.94 Beryllium 9.012 Sodium 22.99 Magnesium 24. In some books, you may see the unit of molar mass as grams/mole or g/mole.I hope you have understood the short and simple calculation for finding the molar mass of Alum. Hence the Molar mass of Alum is 474.372 g/mol. The periodic table lists the atomic mass of carbon as 12.011 amu the average molar mass of carbonthe mass of 6. Now, to calculate the molar mass of Alum, you just have to add the molar mass of all the individual atoms that are present in Alum. The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
